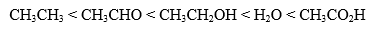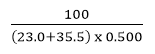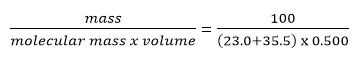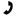In the reaction between NH4+ and NO3–, which of the following would be considered the base?
Increasing Acidity
Which of the following correctly lists the compounds in order of increasing acidity?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
Conjugate Base
The conjugate base of HSO4– is which of the following?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
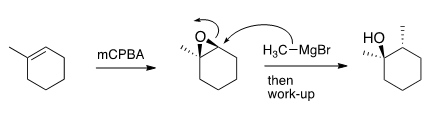
Alkene Identity
An unknown alkene with molecular formula C7H12 undergoes the following reaction to give one product (below). What is the identity of the alkene?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
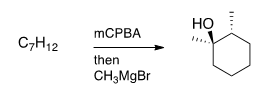
Special Experiment
An experiment is done using a special chemical that includes methylene blue, a compound that turns blue in the presence of oxygen. Three test tubes containing various plant cells exposed to different levels of light are observed over 3 days. Each day the test tube was placed in a photospectrometer and the light transmittance were recorded in the following table.
Which of the following is a possible explanation for the percent transmittance reported for Plant Cell 2?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
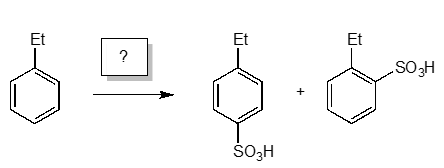
Ethylbenzene Transformation
Which reagents would you use to transform ethylbenzene to the following products?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
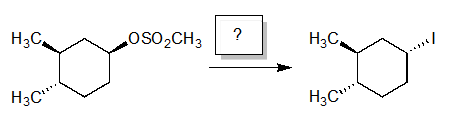
High Yield
Which reagents would you use to perform the following reaction in high yield?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
E2 Reaction
Which is the strongest base for an E2 reaction?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
Buffer Solution
The pH of 1 Molar acetic acid is 2.4. By adding sodium acetate, NaCH3COO to the acetic acid a buffer solution is formed. Which statement best describes what occurs in the buffer solution?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
Molarity of a Solution
The Molarity of a solution formed by dissolving 100g of sodium chloride, NaCl, in 500mL of solution is: