Which of the following is an accurate explanation for evaporative cooling?
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.
Which of the following is an accurate explanation for evaporative cooling?
The solubility of calcium hydroxide (Ca(OH)2) is given as:
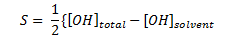
The solubility is a factor of total hydroxide ion in the solution and the solvent hydroxide concentration. The solvent is the liquid in which the calcium hydroxide is dissolved. Which of the following would increase the solubility of calcium hydroxide?
If 7.000g of a solid displaced 21.0 mL of water when dropped into a graduated cylinder, what is the density of the solid?
Consider the following aqueous equation:
6 NaI + Ca3(PO4)2 -> 2 Na3PO4 + 3 CaI2
If 14 moles of NaI, 8 moles of Ca3(PO4)2, and 6 moles of Na3PO4 are present in a mixture, which one of the following compounds would determine the amount of CaI2 that was produced after 2 hours?
When comparing 18O and O-2, which of the following is true?
In the reaction between NH4+ and NO3–, which of the following would be considered the base?
The conjugate base of HSO4– is which of the following?
The pH of 1 Molar acetic acid is 2.4. By adding sodium acetate, NaCH3COO to the acetic acid a buffer solution is formed. Which statement best describes what occurs in the buffer solution?
The Molarity of a solution formed by dissolving 100g of sodium chloride, NaCl, in 500mL of solution is:
In which situation is water acting solely as a Bronsted-Lowry base?