Rank the following aromatic compounds in order of reactivity towards electrophilic aromatic substitution (from most reactive to least reactive):
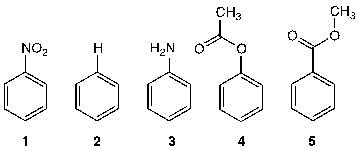

Loading ...
Correct Answer: A. 3 > 4 > 2 > 5 > 1
There are two main electronic effects that substituents can exert: RESONANCE effects include electron donating (e.g. -OMe) where p electrons are pushed toward the arene or electron withdrawing (e.g. -C=O) where p electrons are drawn away from the arene. INDUCTIVE effects include electronegativity type effects. These too can be either electron donating (e.g. -Me) where s electrons are pushed toward the arene or electron withdrawing (e.g. -CF3, -NR3) where s electrons are drawn away from the arene.
Some general pointers for recognizing the substituent effects:
- The H atom is the standard and is regarded as having no effect.
- Activating groups increase the rate (3 > 4)
- Deactivating groups decrease the rate (5 > 1)
Get it right? Get it wrong? Discuss this question with other
DAT Bootcamp students on Facebook:
Discuss on Facebook
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.

