Place the following in order of LEAST acidic to MOST acidic.
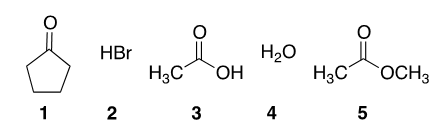

Loading ...
Correct Answer: D. 5 < 1 < 4 < 3 < 2
Mineral acids such as HBr are far more acidic than organic compounds and water; #2 is last in the ranking. From the remaining options, look for trends, and remember the factors that affect the stability of molecules: resonance, charges on atoms with appropriate electronegativities, hybridization and charge.
One way to consider the relative acidities of organic functional groups is to draw resonance structures. Remove the most acidic alpha hydrogen and see how many stable resonance structures you can draw. For instance, carboxylic acids have greater resonance stabilization because of delocalization of the negative charge over two electronegative oxygen atoms.
The alpha hydrogens of ketones (pKa = 20) are more acidic as compared to alpha-hydrogens of esters (pKa = 25); in the ester, there is also a resonance donation from the alkoxy group towards the carbonyl that competes with the stabilization of the enolate charge. This makes the ester enolate less stable than those of aldehydes and ketones so esters are even less acidic.
Get it right? Tweet at us!
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.

