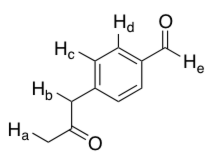
Which labeled proton in the following molecule is most acidic?

Loading ...
Correct Answer: B.
The stability of each resulting conjugate base should be considered; the more stable this is, the stronger the parent acid. For enolates, the greater the degree of charge delocalization (i.e. the more resonance
structures that can be drawn) the stronger the acid. Proton Hb is most acidic due to the charge (and lone pair of electrons) on the conjugate base being delocalized over both carbonyl groups and the benzene ring.
Get it right? Tweet at us!
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.

