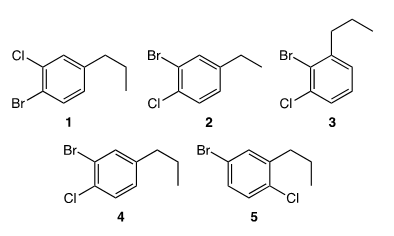
Which of the following molecules corresponds to 2-bromo-1-chloro-4-propylbenzene?

Loading ...
Correct Answer: D. 4
Aromatic compounds are named as derivatives of benzene. Substituents positions are indicated by numbers except that o- (ortho), m- (meta) and p- (para) may be used in place of 1,2-, 1,3-, and 1,4-, respectively, when only two substituents are present. If alkyl substituents are smaller than the ring (6 or fewer carbons), the compound is considered an alkyl substituted benzene. Alkyl substituents larger than the ring (7 or more carbons), are phenyl-substituted alkanes. Compounds are named by numbering the position of each so that the lowest possible numbers are used. The substituents are listed alphabetically when writing the name.
Get it right? Tweet at us!
Subscribe below to get the DAT Question of the Day delivered straight to your inbox every morning.

